1 引言
Poisson-Nernst-Planck 方程, 简称为 PNP 方程, 该方程作为电场和离子浓度分布的耦合方程是描述电扩散过程的一种理想的连续模型[1,2], PNP 方程已被广泛用于研究离子通道、 纳米孔和燃料电池等领域[2⇓⇓-5]. PNP 模型通常假设介电系数是常数, 而实际上, 溶液体系的介电系数与周围的电场、浓度和周围的水化层等都有关系, Booth 等[6]的研究表明, 介电系数是随电场变化的, 这是因为在电解液体系中, 由于强电场的存在, 溶剂分子的偶极子高度定向, 进而影响了溶液体系. Wei 等[7]提出离子的水化作用会影响离子溶液体系的介电系数. 大量实验和理论研究表明, 介电系数是随局部离子浓度的升高而降低的[8,9].
对于介电系数依赖于浓度变化的问题, 文献 [10] 提出了非均匀溶液体系下的 Variable Dielectric Poisson-Nernst-Planck (VDPNP) 方程. 该方程引入了依赖于离子浓度的介电系数, 与经典的 PNP 方程相比, VDPNP 模型在 Nernst-Planck (NP) 方程添加了一个非线性项以及在 Poisson 方程的基础上也考虑了依赖于离子浓度的介电系数, 这个模型能更好地描述电解质溶液中复杂的动力学过程, 但这也给 VDPNP 方程的求解带来了许多困难. 据我们所知, 目前很少有人研究过有关 VDPNP 方程的相关工作, 本文针对 VDPNP 方程, 首先基于有限元方法, 对 VDPNP 模型求解. 其次, 针对纳米孔体系进行数值模拟, 考虑不同电荷密度下, PNP 方程和 VDPNP 方程在纳米孔表面浓度、电势及介电系数分布的模拟结果. 同时, 探讨在混合溶液中改变管壁电荷密度对阳离子分布的影响, 并对比分析 PNP 方程和 VDPNP 方程在纳米孔方向上的阳离子分布特征. 模拟结果表明, 改变管壁的电荷密度对阳离子的分布具有显著影响. 随着电荷密度的增加, 表面效应增强, 导致阳离子的浓度分布发生变化, VDPNP 方程能够更好地捕捉这种电荷密度对阳离子分布的影响, 并且在混合溶液中, 传统的 PNP 方程在浓度上无法有效区分钠离子和钾离子, 而 VDPNP 方程可以明显区分钠、钾离子, 且随着表面电荷越大时, 其区分效果越加明显.
2 VDPNP方程及有限元离散
本文考虑稳态 VDPNP 方程[10]
其中
其中
设
$$\begin{aligned} &H_s^1(\Omega) :=\left\{\phi \in H^1(\Omega): \phi=\phi_0 \text { 在 } \Gamma_D \text{上}\right\}, \\ &H_{s, 0}^1(\Omega) :=\left\{v \in H^1(\Omega): v=0 \text { 在 } \Gamma_D \text{上}\right\}. \end{aligned}$$
令
$$\int_{\Omega}-\nabla \cdot(\epsilon(\bar{c}) \nabla \phi) v{\rm d}V=\int_{\Omega} \rho(c) v{\rm d}V, \quad \forall v \in H_{s, 0}^1(\Omega).$$
令
为了得到 (2.1) 式的变分形式, 定义
$$\begin{aligned} V & :=\left\{c_i\left|c_i \in H^1\left(\Omega_s\right): c_i\right|_{\partial \Omega}=c_i^{\infty}\right\}, \\ V_0 & :=\left\{w\left|w \in H^1\left(\Omega_s\right): w\right|_{\partial \Omega}=0\right\}. \end{aligned}$$
求
寻找
区域
$$V_h=\left\{v \in H^1(\Omega):\left.v\right|_{\partial \Omega}=0, \left.v\right|_e \in P(e), \forall e \in \mathcal{T}_h\right\},$$
则 (2.1)-(2.2) 式的标准有限元离散为: 对
3 数值实验
本节给出数值例子验证 VDPNP 模型的可求解性. 实验环境为桂林电子科技大学超算平台 (Linux), 内存为 3.58 TB, 采用 For
下面以二维为例, 考虑如下只有正、负两个离子的 VDPNP 模型, 即正离子浓度
$$\begin{aligned} &-\nabla \cdot\left(\nabla p+p \nabla\left(\phi-\frac{1}{2} \epsilon^{\prime}(\bar{c})|\nabla \phi|^2\right)\right) =F_1, & \text { 在 } \Omega_s \text{上},\\ &-\nabla \cdot\left(\nabla n-n \nabla\left(\phi-\frac{1}{2} \epsilon^{\prime}(\bar{c})|\nabla \phi|^2\right)\right) =F_2, & \text { 在 } \Omega_s \text{上}, \\ &-\nabla \cdot(\epsilon(\bar{c}) \nabla \phi)-(p-n) =F_3, & \text { 在 } \Omega_s \text{上}, \end{aligned}$$
右端函数依赖于解析解, 其中解析解定义为
VDPNP 方程是一类非线性耦合方程组, 常用的解耦方法是 Gummel 迭代方法, Gummel 迭代方法不仅具有较快的求解速度、良好的收敛性和稳定性, 还可以根据实际需求灵活调整迭代次数和收敛准则, 从而达到所需的数值解精度, 本文 Gummel 迭代如算法 1 所示. 令
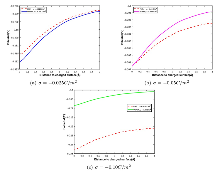
在计算中, 选取不同空间步长
表1 VDPNP 精确解和有限元解的
由表 1 可知, 在
4 VDPNP方程的微纳米孔系统数值模拟
对于生物分子周围局部高浓度的溶液, 考虑如下的非线性介电模型[11]
其中
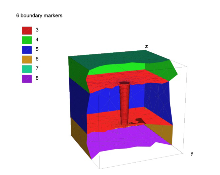
考虑用纳米管数值模拟 VDPNP 对体系相互作用的影响, 将要模拟的网格示意如图1 所示, 其中外层正方体盒子的边长为
图1
图1
纳米管模拟网格示意图.
注:
我们考虑 1 : 1
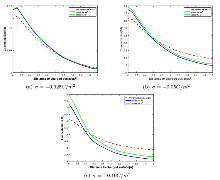
由图2 的模拟结果表明, PNP 与 VDPNP 模拟在管壁表面附近的阳离子分布存在差异, 且随着表面电荷越大, 差异越显著; 当表面电荷较低时 (如图2(a) 所示), VDPNP 模拟的阳离子浓度高于 PNP; 另外图2 的 (b), (c) 表明, 当带电表面的电荷增大时, VDPNP 模拟的结果并不再始终大于 PNP, 在离管壁的一定距离处, VDPNP 模拟出的阳离子浓度低于 PNP, 且随着电荷增大, VDPNP 的阳离子浓度在靠近纳米孔表面比 PNP 的要高, 产生这种物理现象的原因可能是由于介电系数的引入使得 VDPNP 方程可能更细致地考虑了离子与带电表面之间的相互作用,如静电吸引和排斥, 这些相互作用会导致阳离子在靠近表面时更紧密的吸附, 浓度增加.
图2
电势的模拟结果如图3 所示, 在低电荷下, VDPNP 模型模拟的电势低于 PNP 模型, 高电荷下 VDPNP 模型的电势高于 PNP. 随着表面电荷密度的增加, 两者的电势差异逐渐增大.
图3
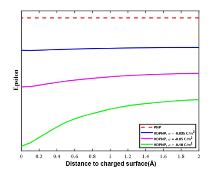
介电系数分布结果如图4 所示, 表面电荷密度越大, 介电系数沿管壁的径向分布变化越显著. 同时, 表面电荷密度越高, 管壁附近的阳离子浓度越大, 介电系数越小, 与介电模型相符.
图4
下面我们进一步研究了纳米孔体系中
图5
图6
5 结论
本文基于有限元方法对 VDPNP 模型进行了求解, 数值误差结果验证了方程的可解性和数值程序的稳健性. 通过对纳米孔数值模拟, 结果表明, 在纳米孔体系中, 传统的 PNP 方程在浓度上无法有效区分钠离子和钾离子, 而 VDPNP 模型则能够明显区分这两种离子, 并且随着表面电荷密度的增加, 离子区分效果愈加显著. 这表明 VDPNP 模型能够更精确地捕捉复杂电荷相互作用和浓度梯度效应, 适用于纳米尺度下的离子传输和选择性识别问题. 研究结果为理解电荷调控和离子选择性提供了新的理论依据, 并为纳米技术和电化学传输等领域的进一步研究提供了参考.
参考文献
Poisson-Nernst-Planck equations for simulating biomolecular diffusion-reaction processes I: Finite element solutions
In this paper we developed accurate finite element methods for solving 3-D Poisson-Nernst-Planck (PNP) equations with singular permanent charges for electrodiffusion in solvated biomolecular systems. The electrostatic Poisson equation was defined in the biomolecules and in the solvent, while the Nernst-Planck equation was defined only in the solvent. We applied a stable regularization scheme to remove the singular component of the electrostatic potential induced by the permanent charges inside biomolecules, and formulated regular, well-posed PNP equations. An inexact-Newton method was used to solve the coupled nonlinear elliptic equations for the steady problems; while an Adams-Bashforth-Crank-Nicolson method was devised for time integration for the unsteady electrodiffusion. We numerically investigated the conditioning of the stiffness matrices for the finite element approximations of the two formulations of the Nernst-Planck equation, and theoretically proved that the transformed formulation is always associated with an ill-conditioned stiffness matrix. We also studied the electroneutrality of the solution and its relation with the boundary conditions on the molecular surface, and concluded that a large net charge concentration is always present near the molecular surface due to the presence of multiple species of charged particles in the solution. The numerical methods are shown to be accurate and stable by various test problems, and are applicable to real large-scale biophysical electrodiffusion problems.
Poisson-Nernst-Planck (PNP) theory of an open ionic channel
Poisson-Nernst-Planck model of ion current rectification through a nanofluidic diode
Variational multiscale models for charge transport
DOI:10.1137/110845690 URL [本文引用: 1]
Ionic channels in biological membranes: Natural nanotubes
DOI:10.1021/ar950051e URL [本文引用: 1]
Dielectric constant of polar liquids at high field strengths
DOI:10.1063/1.1742009
URL
[本文引用: 1]

A critical examination is given of a simplifying assumption made in an earlier calculation of the functional dependence of the integral dielectric constant of polar liquids on the applied field. It was assumed in this calculation that, for the cavity and reaction fields at all field strengths the usual expressions for low field strengths could be employed. It was pointed out that this simplification is plausible if the static dielectric constant is large compared with the optical dielectric constant. In the present note it is shown that, if it is postulated that the reaction field alone retains its conventional value, then the cavity field is appreciably increased by dielectric saturation. In the case of water the effect is equivalent to an increase of the external field by about 10 percent; a modified relation between the field and the dielectric constant is suggested.
Ion size effects on the dynamic and static dielectric properties of aqueous alkali solutions
DOI:10.1063/1.462792
URL
[本文引用: 1]

Dielectric spectroscopy studies of aqueous ionic solutions ACl/H2O (A=Li, Rb, Cs) were carried out at frequencies between 45 MHz and 20 GHz, using recently developed coaxial measurement techniques. The behavior of the static dielectric constant εs0 and the dielectric relaxation time τD of the solutions were studied as functions of ion size and concentration. For moderate concentrations both εs0 and τD decrease linearly with solution conductivity. While the behavior of εs0 can be understood in terms of either static or Hubbard–Onsager kinetic polarization models, the experimental results for τD are at present not understood quantitatively in terms of these models. However we point out the good correlation of the τD data with empirical viscosity results, which suggests an alternative explanation based upon the solution viscosity, modified by ion size effects, which play an important role in the dielectric response. We also discuss the various length scales relevant to dielectric and conductivity processes in the solutions.
Modeling multibody effects in ionic solutions with a concentration dependent dielectric permittivity
Mean-field theory and computation of electrostatics with ionic concentration depenent dielectrics
We construct a mean-field variational model to study how the dependence of dielectric coefficient (i.e., relative permittivity) on local ionic concentrations affects the electrostatic interaction in an ionic solution near a charged surface. The electrostatic free-energy functional of ionic concentrations, which is the key object in our model, consists mainly of the electrostatic potential energy and the ionic ideal-gas entropy. The electrostatic potential is determined by Poisson's equation in which the dielectric coefficient depends on the sum of concentrations of individual ionic species. This dependence is assumed to be qualitatively the same as that on the salt concentration for which experimental data are available and analytical forms can be obtained by the data fitting. We derive the first and second variations of the free-energy functional, obtain the generalized Boltzmann distributions, and show that the free-energy functional is in general nonconvex. To validate our mathematical analysis, we numerically minimize our electrostatic free-energy functional for a radially symmetric charged system. Our extensive computations reveal several features that are significantly different from a system modeled with a dielectric coefficient independent of ionic concentration. These include the non-monotonicity of ionic concentrations, the ionic depletion near a charged surface that has been previously predicted by a one-dimensional model, and the enhancement of such depletion due to the increase of surface charges or bulk ionic concentrations.
Analysis of the mean field free energy functional of electrolyte solution with nonhomogenous boundary conditions and the generalized PB/PNP equations with inhomogeneous dielectric permittivity
DOI:10.1137/16M1108583 URL [本文引用: 2]
An ionic concentration and size dependent dielectric permittivity Poisson-Boltzmann model for biomolecular solvation studies
Ionic size effects: Generalized Boltzmann distributions, counterion stratification, and modified Debye length
Near a charged surface, counterions of different valences and sizes cluster; and their concentration profiles stratify. At a distance from such a surface larger than the Debye length, the electric field is screened by counterions. Recent studies by a variational mean-field approach that includes ionic size effects and by Monte Carlo simulations both suggest that the counterion stratification is determined by the ionic valence-to-volume ratios. Central in the mean-field approach is a free-energy functional of ionic concentrations in which the ionic size effects are included through the entropic effect of solvent molecules. The corresponding equilibrium conditions define the generalized Boltzmann distributions relating the ionic concentrations to the electrostatic potential. This paper presents a detailed analysis and numerical calculations of such a free-energy functional to understand the dependence of the ionic charge density on the electrostatic potential through the generalized Boltzmann distributions, the role of ionic valence-to-volume ratios in the counterion stratification, and the modification of Debye length due to the effect of ionic sizes.
A local approximation of fundamental measure theory incorporated into three dimensional Poisson-Nernst-Planck equations to account for hard sphere repulsion among ions
DOI:10.1007/s10955-016-1470-7 URL [本文引用: 1]